World Fire Fuel List
Isopar
Isopar Performance
Isopar Availability
Isopar Safety
Comments from an analytical chemist
Safety of Colemans Fuel
Fire Fuel Assays
Additional Downloads
Hey there! My name is Dan Miethke. Fire has been my passion for close to a quarter century. I’ve worked as a fire performance professional for over 20 years including consulting for Cirque du Soleil and Franco Dragone during the creation of fire acts.
I’ve performed in over 60 countries and had to research international fuel availability and safety, talking to local performers and more recently associates with a vastly higher applicable education (Big thanks to Matt ‘Silence’ Sweeney, Ed Hollings, Arashi and many more).
I’ve been compiling a worldwide fire fuel list with flash points, manufacturers and sources. I’m sharing this in the hopes that other professional and recreational fire artists will find it useful and to promote a deeper understanding of the fuels we burn. Perhaps it can play a part in reducing unnecessary risks, smells and soot in the industry as a whole and fire jams worldwide.
If anyone wants to contribute or make suggestions please email me via dan@sparkfiredance.com
World Fire Fuel List: http://bit.ly/worldfirefuels
ISOPAR
My personal opinion is that the safest selection of fuels regarding exposure/toxicity in liquid/vapour form are the odourless isoparrafin range marketed as ISOPAR by ExxonMobil. All suitable for indoor use.
ISOPARs are available in a range of flash points.
ISOPAR C -8c
ISOPAR E 7c
ISOPAR G 41c
ISOPAR H 54c
ISOPAR L 64c
ISOPAR M 93c
ISOPAR PERFORMANCE
For the purposes of fire performance ISOPAR E and ISOPAR G should cover most common situations or effect requirements.
ISOPAR C might be useful in very cold climates or for fast evaporation (flame effects in water shows) though with the lower flash point has increased storage risks. ISOPAR L can be used in situations where a combustible fuel (flash point greater than 61c) rather than a flammable fuel is necessary for storage/legal/OH&S requirements.
ISOPAR E will burn/react similar to Colemans, Zippo Lighter Fluid, Essence F etc. ‘Low flash point fuels’.
Lighting very quickly, evaporating off props after fuelling, not causing slippery stages, burning on costumes for a few seconds with a heavy prop hit, requiring quite some extra safety precautions in handling, transport and on site.
ISOPAR G will burn/react fairly similar to Lamp oil, Paraffin, Shellsol T, Kerdane etc. ‘High flash point fuels’.
Longer lasting, takes a second or two to light, spray off will be a little slippery, wont burn on you if you hit yourself, vapours are not a serious fire hazard during fuelling, much safer to transport and store.
Some advantages of ISOPAR G to the other fuels mentioned, it will evaporate off props eventually. After a performance most remaining fuel will evaporate off hot props fairly quickly leaving an odourless almost clean prop for transport. (Some of the very last traces can take a day or so in the open for complete evaporation to leave the prop unlightable for flights etc.)
ISOPAR G will also evaporate off stage surfaces, but rather than in the few seconds it would take ISOPAR E, Colemans etc. it will take ISOPAR G 30sec to a few minutes before a fine stage spray will disappear.
A combination of ISOPAR G and a lower flash point fuel can make working on dance floors rather than carpet practical, and with eventual evaporation is less of an environmental load if performing outdoors.
ISOPAR AVAILABILITY
The ISOPARs are available in 20L drums from some distributors in some countries, i.e. Australia, but in most countries a 200L drum purchase will be required.
There will be other substitutes for ISOPARs made by regional companies (IP155 by Total Fluids in France) but ISOPARs are well known and used in various industries worldwide.
This makes it worthwhile for long term recreational or domestic performance use. For international performances alternatives will often be required though it is worth noting that various ISOPARs are sold as professional fire fuels by juggling stores and online fire toy retailers and these can often be shipped to your gig in advance.
ISOPAR SAFETY
A Chemical Exposure Monitoring Report was conducted for the fire act I was performing during my years at Cirque. Personal exposure monitoring of the artistes and technicians was conducted with body mounted devices. The main fuel used was ISOPAR G.
All the personal exposure readings were less than the occupational exposure limits, except for short periods during prop extinguishing.
The report indicates the highest exposures are when props are ‘spun off’ or extinguished, still hot with the fuel vaporising but not combusting. This period holds the largest risk of inhaling a high amount of fuel vapour.
For fuelling I recommend a clear squeeze bottle with measured amounts of the fuel (applicator measuring bottle). This measurement is quite quick to figure out for each prop and eliminates the need for a spin or squeeze off. With a little practice this can also eliminate stage spray off, reducing slip hazards. Start with 25ml for small wicks, 75ml for large wicks.
For extinguishing leave your props wrapped in wet towels after extinguishing and take them somewhere ventilated to air out after a show. Leaving a used prop wrapped during the rest of a performance is also good standard practice to avoid a vapour re-ignition.
I have read various reports on isoparrafin exposure over the years and they all seem to indicate quite high levels of exposure to liquids/vapours are needed for serious effects, with most being reversible on cessation of exposure.
A separate and much harder to quantify hazard is the remaining components of incomplete combustion of fuel, the soot and smoke. See more on that below.
Personally I maintain movement in most routines designed for indoor use, avoiding long pauses with static burning props (laminar flames). I find this can greatly reduce the amount of soot/smoke from a fire act as the flames get a little more oxygen with the movement and become turbulent flames, burning a touch more efficiently.
COMMENTS FROM AN ANALYTICAL CHEMIST (Matt ‘Silence’ Sweeney)
“Right, so I think everyone has a pretty good understanding of the toxicity risks of liquid fuels -before- they are burned; with benzene being an exemplary compound for a cyclic aromatic, and will all/most cyclic aromatics (and most anything with delocalised pi orbitals, if you want to get technical) being generally carcinogenic / teratogenic / etc.. So in that overly simple sort of analysis, then the highly purified alkanes (e.g., paraffin oils and some of the trade names suggested) are relatively free of such compounds (but not as free of them as bio diesel, veg oil, or alcohols – without getting into the suitability as a firespinning fuel).
However, looking at MSDS sheets is of basically zero use for determining the toxicity of the (incomplete) combustion products of a fuel. Now, the vapour one inhales from spinning also includes vaporized and uncombusted fuel, so the dangers of the liquid fuel are relevant … but that also requires one to look at the additional variable of clearance from the body. So, for example, coleman fuel is pretty toxic, but if you drink it or inhale a bunch of it, you will breath it out over time because it evaporates so readily. Whereas ultrapure paraffin oil is rather non-toxic, but if you inhale a mist/vapour of it it has a high boiling point and will stay inside you body much longer, in the case of firebreathing chemical pnemonia is usually caused by a mist of non-evaporating oil coating the lungs and causing an immune reaction (the fuel doesn’t have to even be toxic, just a foreign material to the body). As immune responses are what they are, this is why some people become allergic/sensitized over time to paraffin oil (lamp oi / kerosene) smoke, but almost nobody is allergic to coleman, regardless of how sooty and gross it can be. That isn’t to say safer, just different risks.
Now let us consider what happens to fuel when it burns – and I don’t mean when it burns completely to H2O and CO2, because then it would have a weak blue flame like ethanol on the wicks and would not be a pretty firespinning fuel. Move around on the carbon chain, and things get highly reactive (I could go into more detail, but I’ll stop), and these mobile reactive parts of the carbon chain are somewhat random in their movement and reactivity. So the longer (and more branched or cyclic) the fuel molecule, the greater the chance that the radical intermediaries are going to react with something (other than O2 or some other O compound) and form something ‘new’. These new things can be incredibly toxic. So for example, methane (CH4) burns pretty cleanly, while a triglyceride (say, bacon fat which is 3 18 carbon chains with a glycerol linker, C55H98O6) will have a lot more chances to make fun new things.
This incomplete combustion and side product formation is pretty easy to predict; basically all of the red/yellow/orange color from fire is glowing soot (I like to stay away from the p word), whereas complete combustion emits very little light an almost no color (think a bunsen burner).
Ok, so anything that burns with (fire performance usable) color is basically toxic/carcinogenic. But let us get back to the evaporation point question. The incomplete combustion products of a lower boiling point fuel (coleman) are going to be a mix of boiling points, but those are going to be much lower, on average, than the boiling points of highly pure paraffin oil. So the clearance from the body is going to be similarly affected, with the chance of long term exposure in the lungs being much higher for, say, burning olive oil (which is totally non toxic unburnt), that it would be for methanol, as methanol burns rather cleanly even if it is super toxic as a liquid.”
“Did the Cirque carbon black testing cover all the potential nasties from incomplete combustion or do we still lack data for other products such as polycyclic aromatic hydrocarbons?”
Matt: Probably not. My analytical chemistry background is in this general area, and doing this work in very difficult. I can imagine getting an estimate, but the problem is, even with a perfect detection setup (which almost nobody would have access to) I can’t really imagine that they would replicate burn/fume conditions very well for all possible scenarios. Changes in air temp, pressure, humidity, etc, on top of prop type, wick condition, spin rate, etc., sort of make this impossible to do as more than an estimate, even for just one very pure fuel.
How much higher would carbon black / PAH (particle-bound polycyclic aromatic hydrocarbons) exposure be for the longer carbon chain, higher flashpoint fuels?
Matt: Much higher, I’m fairly certain of that.
Does the lower boiling point and faster clearance of the low flash point fuels negate the concerns of exposure to benezene, toulene and n-hexane?
Matt: Now this one is a hard question to answer, at least without testing on a bunch of mice or something extreme, but I will hazard an educated guess and say “yes, probably,” and I select my fuels as such. Empirically, I know almost nobody who has developed allergies or sensitivity to the lighter fuels, but we all know people who can’t be around kero or lamp. Also, getting the stuff stuck in your lungs applies more stress to one area, vs a systemic toxicity. This is worth further study; that said, reducing the cyclics in the lighter fuels is absolutely a good thing to aim for.
– What levels of benzene exposure could we be exposed to? (for example when using Zippo which is 2% benezene and 7% toulene)
Matt: Sadly if it is an unregulated product, this will almost certainly vary from batch to batch. Kerosine in the US can be all over the place, for instance (while kerosine in europe is usually pretty clean, relatively speaking). Benzene is incredibly carcinogenic…
– How do we mitigate this exposure apart from the obvious i.e. good ventilation?
Properly spun off wicks, doing that in an area where people will not be breathing to spun off fuel, lighting (or bagging) props quickly. Masks won’t help (much or at all) unless they are the super serious ones painters use, those pollution masks are not going to stop gasses.
Thanks Matt!!
SAFETY OF COLEMANS CAMP FUEL
Colemans fuel is used widely in the USA. Unfortunately with branded retail products the exact composition is considered proprietary in some countries and manufacturers do not need to release specific data sheets.
This, combined with the fact that retail camp fuel companies can change the formula at any time without notice (and regularly do, trying to create a consistent product out of inconsistent crude sources) makes it hard to know how much of a dangerous chemical such as Hexane is in the product.
The following are excerpts from a couple of reports which give some cause for concern based on the n-hexane contents, though they seem to lump colemans in with various other ‘white gas’ camp fuel brands in the US.
“Coleman Fuel is representative of a class of commercially available petroleum-based products that are often marketed under the following names: Amsol 10, Kensol 10, White Gas, VM&P Naphtha, Ozark Trail, Camp Fuel, and Coleman Fuel (brand).“
“These products are complex mixtures of light hydrotreated petroleum distillates containing up to 25% n-hexane, 15% cyclohexane, and less than 0.001% benzene by weight.” (*these percentages need clarification, sourced from just two reference texts (1) (2) seemingly referring to a wide range of low flashpoint fuels. See Fuel Assays below for mass spectrometer analysis)
“Repeated exposure to Coleman Fuel and its constituents, such as n-hexane, over a period of weeks to months can result in neuropathy, damage to sensory and motor nerve cells. Effects include burning, numbness, or tingling in the feet, legs or hands, paralysis, and even permanent brain damage. Prolonged exposure may also damage the kidneys, liver, and respiratory system.”
“The 10-hour Time-Weighted-Average Occupational Exposure Limit for n-hexane, which comprises up to 25% of Coleman Fuel, is 50ppm (180 mg/m3). The 8-hour Time-Weighted-Average for n-hexane is 500 ppm (1800 mg/m3).”
More info on Hexane here: http://www.toxipedia.org/display/toxipedia/Hexane
It’s unfortunate we cannot get specific data on Colemans. Heptane would be a safer alternative to Hexane (as used in Swedens Primus Powerfuel) but is probably a bit more expensive. In the end with an allowance of up to 25% hexane it’s left to the user to guess whether colemans is prioritising their customers health, or their profit.
Although I have used, and will personally occasionally use Colemans again I believe ongoing exposure from repeated long term recreational or professional use may pose unnecessary risks due to the Hexane content, with safer low flash point substitutes such as Isopar E available.
Other low flash point fuels have their own concerning ingredients. Zippo having up to 2% Benzene allowance in some of it’s lower quality versions is of personal concern as it’s a great last resort for shows in odd locations.
On the plus side for all the low flash point fuels see Matts comments above regarding faster clearance times from the body of the faster evaporating fuels.
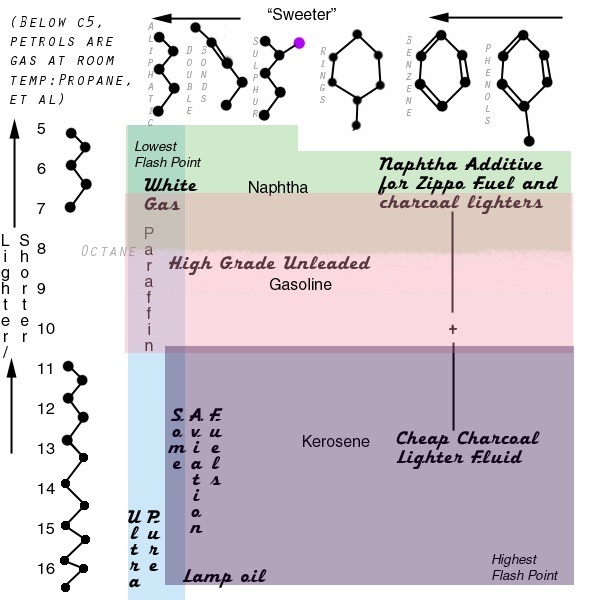
FIRE FUEL ASSAYS
Source: http://ilrc.ucf.edu/sample_detail.php?sample_id=35
ISOPAR E
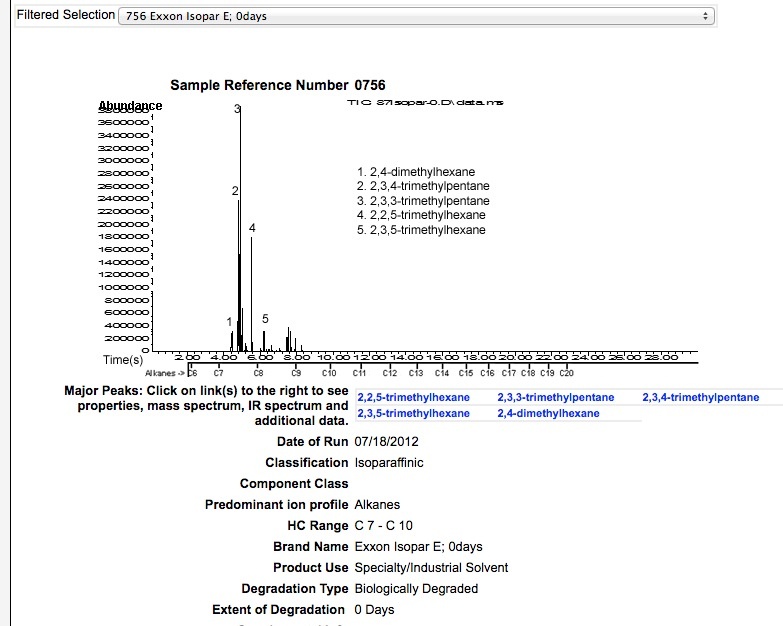
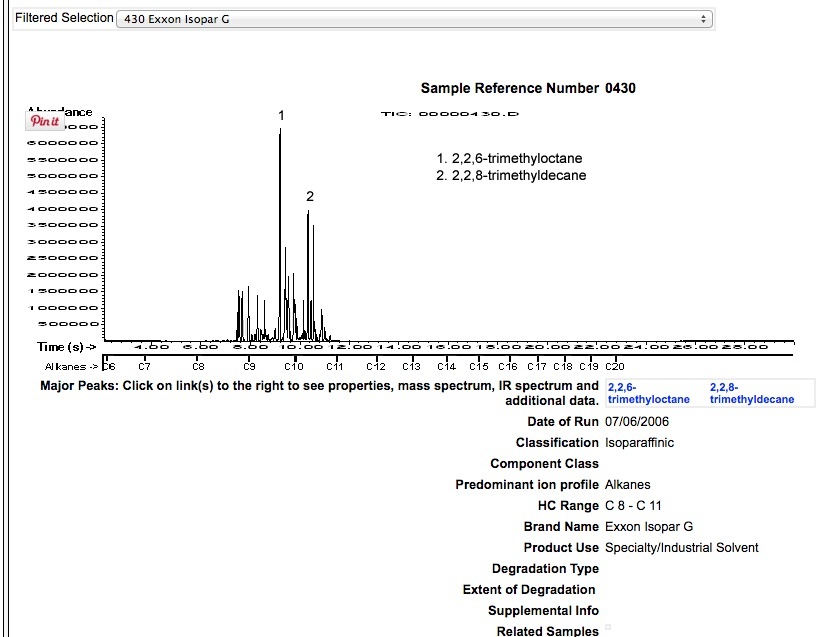
ISOPAR G
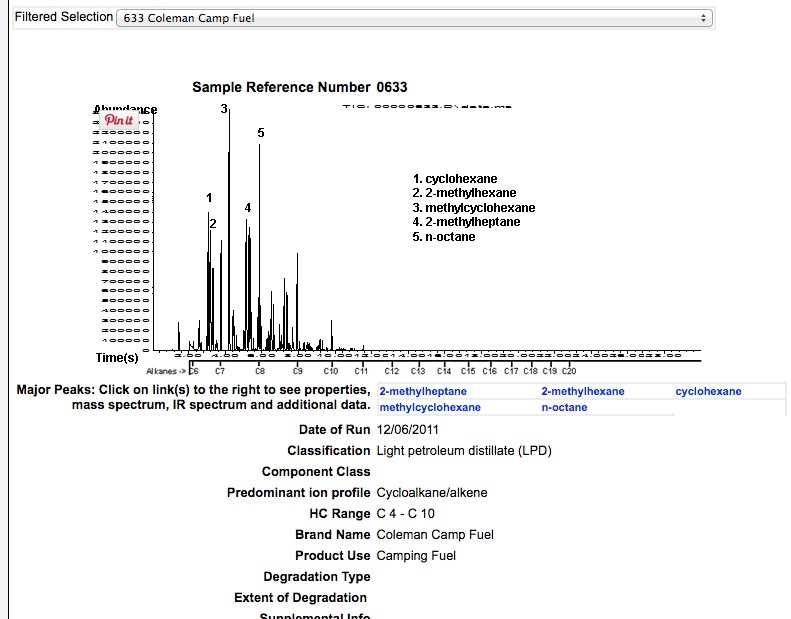
COLEMANS
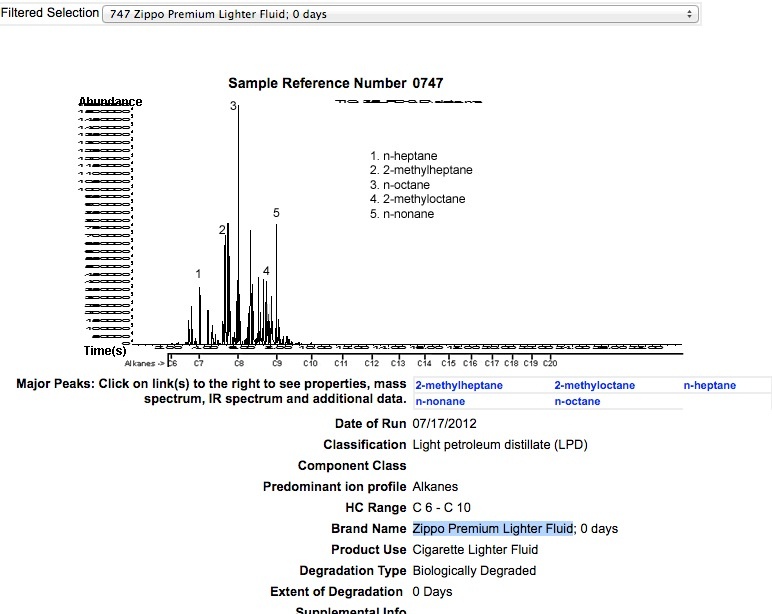
ZIPPO PREMIUM LIGHTER FLUID
ULTRA PURE LAMP OIL (UPLO)
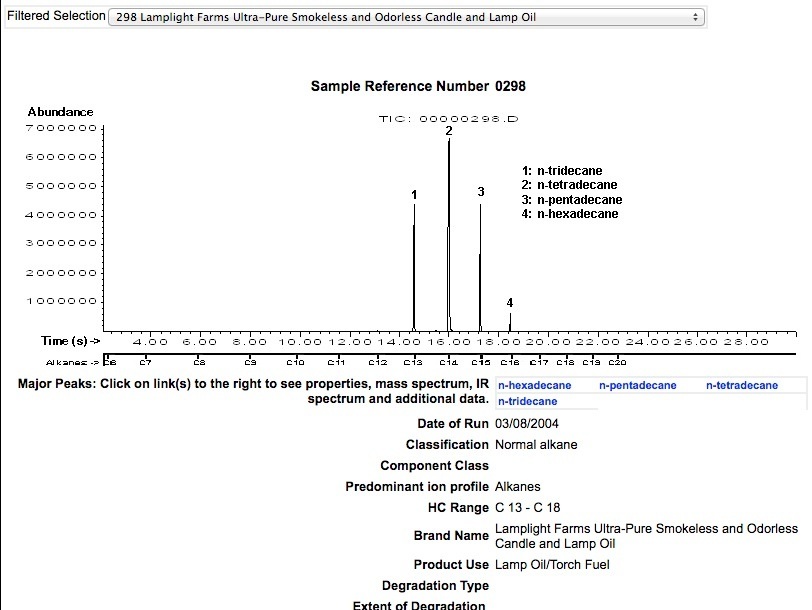
PARAFFIN

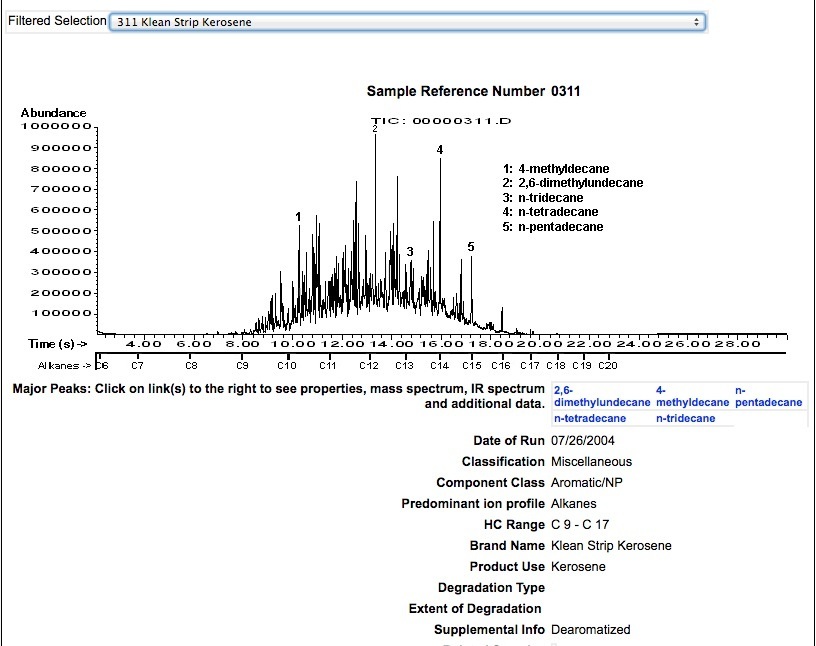
KEROSENE
ADDITIONAL FILES OF INTEREST
Relevant partial summary of cirque atmospheric testing data
C10-11 Isoparaffins used in Cosmetics
Painters Carcinogenic info 20%-40% cancer above average
Flame Heat Trials – Colemans – UPLO – Alcohol – timed temperatures
Thermal properties of different Fibres

- Spark Fire Dance on ‘Das Supertalent’ – RTL - September 28, 2016
- How to choreograph a circus act? - September 28, 2016
- Scientists just discovered a new type of eco-friendly fire - August 8, 2016



